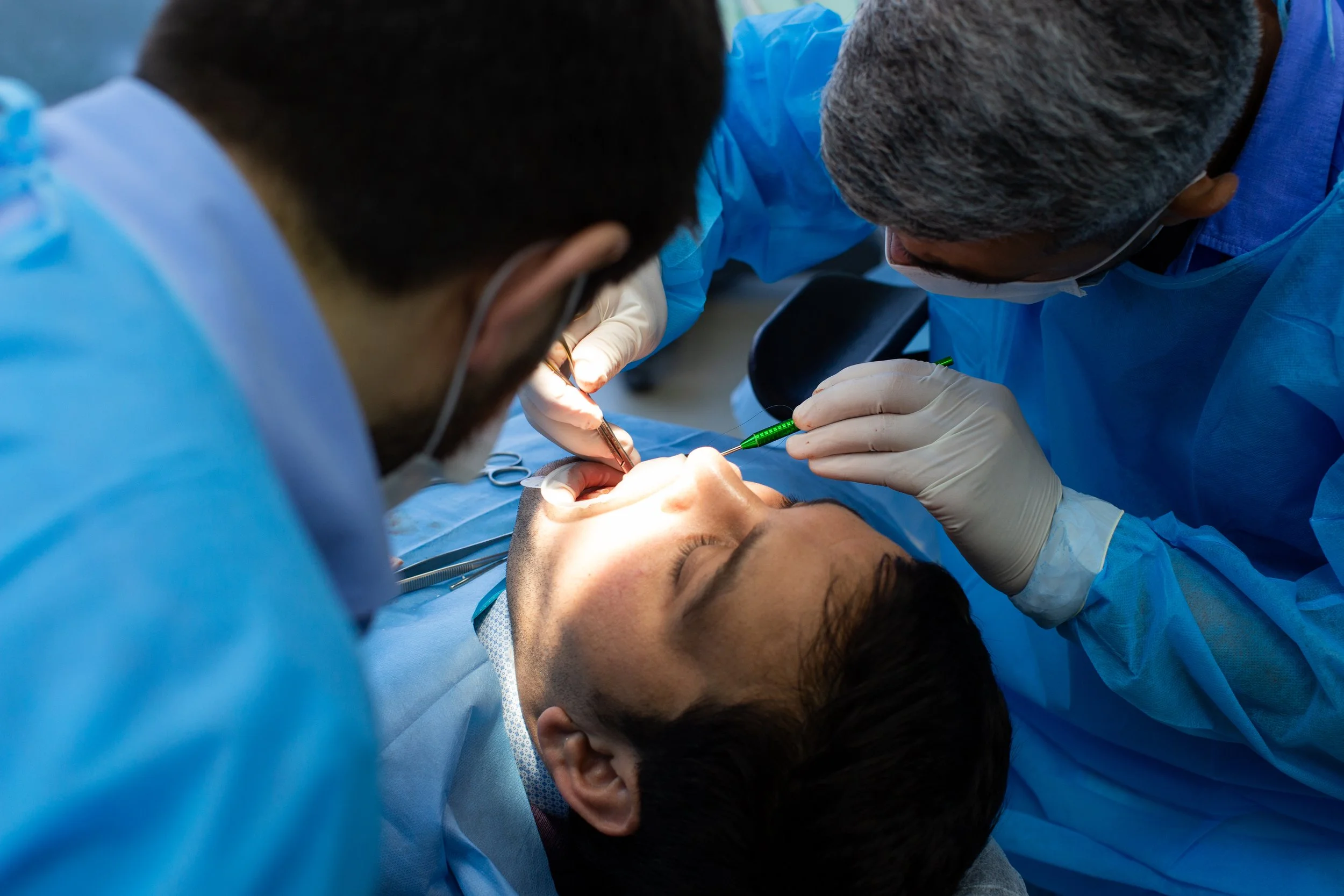
Clinical Applications
Minor Bone Augmentation
Our bone substitutes are ideal for small to moderate defects: peri-implant gaps, small ridge contouring, localised sinus elevations, periodontal regeneration.
Benefits: Easy to handle and adapt, supports early vascularisation, predictable integration and volume stability.
Major Bone Augmentation
For extensive ridge defects, our biomaterials deliver both scaffold support and biological. performance: horizontal & vertical ridge reconstruction, severe atrophy cases before implant placement, combined GBR with membrane stabilisation.
Benefits: Cortical strength for space maintenance, cancellous porosity for cell ingrowth, compatible with tenting screws, meshes or sausage-technique.
Socket Management
Maintain ridge volume following tooth extraction with reliable, biologically active graft materials — preventing resorption and supporting natural healing.
Benefits: Maintains alveolar dimensions, promotes natural bone replacement, simplifies later implant placement, works synergistically with resorbable membranes.
The “Sausage Technique”
Our materials are perfectly suited for this minimally invasive GBR method: bone grafts stabilised beneath a membrane using sutures or tacks to create tension that shapes and maintains graft volume.
Benefits: High graft stability, predictable dimensional bone gain, reduced surgical morbidity, excellent soft-tissue adaptation.
Other Regenerative Indications
Our products suit Sinus lift (lateral & crestal), ridge split/expansion procedures, peri-implantitis defect repair, cystic cavity filling, periodontal intrabony defects, soft-tissue contour enhancement.
Benefits: Text needed here Text needed here Text needed here Text needed here Text needed here Text needed here Text needed here.
Technical Summary & Advantages.
Our biomaterials are designed to support predictable regenerative outcomes through a combination of robust processing, clinically useful formats, & dependable supply.
From terminal gamma irradiation that balances safety with preservation of tissue structure, to a broad set of particle size options that let clinicians match the graft to defect morphology and technique, the range is built for real-world surgical decision-making.
UK-based processing and distribution helps simplify logistics, strengthen traceability, and improve consistency of supply for practices and groups. As human allografts, these products support biologic compatibility while avoiding donor-site morbidity associated with autogenous harvesting.
Taken together, hard and soft tissue options under one portfolio - this creates a practical, streamlined approach that can reduce complexity in procurement, training, and day-to-day clinical workflows.
-
Allograft tissues processed and sterilised via gamma irradiation at 15-25 kGy — terminal sterilisation ensures safety, meets industry standard sterilisation dose benchmarks while preserving tissue structure and performance. (Industry literature supports gamma irradiation as optimal for maintaining biocompatibility in pericardial/dermal scaffolds)
-
For mineralised cortical-cancellous bone powder we offer multiple granule/particle size ranges (150-500 μm, 150-1000 μm [most popular], 500-1000 μm, 1000-2000 μm, 150-2000 μm) enabling clinicians to tailor choice to defect morphology and surgical technique.
-
Being processed in the UK helps reduce logistic complexity, simplifies regulatory/import concerns, ensures traceability and supports clinicians focused on supply-chain reliability and UK-based sourcing.
-
Using human allograft (rather than xenograft) reduces donor-site morbidity, enhances biological compatibility and supports predictable regeneration.
-
With options across hard-tissue (bone powders) and soft-tissue (membranes/grafts) regeneration, clinicians can use a single supplier for multiple indications, simplifying procurement and training.
Resources
Adverse Events
Clinical Evidence
Safety & Quality

